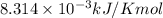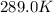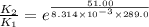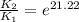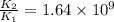Chemistry, 01.04.2020 22:23 milkshakegrande101
The presence of a catalyst provides a reaction pathway in which the activation energy of a reaction is reduced by 51.00 kJ ⋅ mol − 1 51.00 kJ⋅mol−1 . Uncatalyzed: A ⟶ B A⟶B E a = 136.00 kJ ⋅ mol − 1 Ea=136.00 kJ⋅mol−1 Catalyzed: A ⟶ B A⟶B E a = 85.00 k J ⋅ mol − 1 Ea=85.00 kJ⋅mol−1

Answers: 1
Another question on Chemistry



Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2

You know the right answer?
The presence of a catalyst provides a reaction pathway in which the activation energy of a reaction...
Questions

History, 09.11.2020 23:40

Mathematics, 09.11.2020 23:40





English, 09.11.2020 23:40



Social Studies, 09.11.2020 23:40

Mathematics, 09.11.2020 23:40



World Languages, 09.11.2020 23:40

Mathematics, 09.11.2020 23:40


Mathematics, 09.11.2020 23:40

Arts, 09.11.2020 23:40


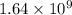
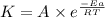
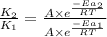
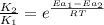
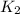 = rate of reaction with catalyst
= rate of reaction with catalyst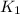 = rate of reaction without catalyst
= rate of reaction without catalyst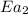 = activation energy with catalyst
= activation energy with catalyst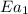 = activation energy without catalyst
= activation energy without catalyst