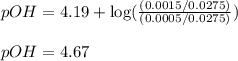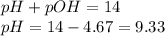Chemistry, 01.04.2020 22:34 raprocksbob
Calculate the pH during the titration of 20.00 mL of 0.1000 M trimethylamine, (CH3)3N(aq), with 0.2000 M HCl(aq) after 7.5 mL of the acid have been added. Kb of trimethylamine = 6.5 x 10-5.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:00
1)each group 16 element has how many valence electrons? ( )4 ( )6 ( )8 ( )16 2)how many dots appear in the dot structure for calcium ion, ca2+? ( )zero ( )one ( )two ( )eight 3) which of the following atoms forms a cation to obtain an octet of outer shell electrons? ( )magnesium ( )oxygen ( )fluorine ( )helium 4) an al3+ ion contains 13 protons and 10 electrons. ( )true ( )false 5) valence and non-valence electrons are represented in lewis dot structures. ( )true ( )false
Answers: 3

Chemistry, 22.06.2019 07:50
Which of the following electromagnetic waves can create ions?
Answers: 2

Chemistry, 22.06.2019 15:30
Which suspect most likely committed the robbery and how do you know
Answers: 2

Chemistry, 22.06.2019 20:30
What is a difference between a mixture of elements and a mixture of compounds
Answers: 1
You know the right answer?
Calculate the pH during the titration of 20.00 mL of 0.1000 M trimethylamine, (CH3)3N(aq), with 0.20...
Questions

Chemistry, 09.01.2020 14:31

Mathematics, 09.01.2020 14:31




Spanish, 09.01.2020 14:31




Mathematics, 09.01.2020 14:31




Mathematics, 09.01.2020 14:31

History, 09.01.2020 14:31

Mathematics, 09.01.2020 14:31


Mathematics, 09.01.2020 14:31

Mathematics, 09.01.2020 14:31

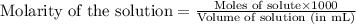 .....(1)
.....(1)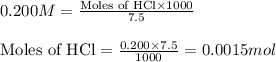
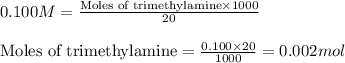
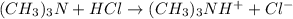
![pOH=pK_b+\log(\frac{[\text{conjugate acid}]}{[\text{base}]})](/tpl/images/0576/5686/2dffc.png)
![pOH=pK_b+\log(\frac{[(CH_3)_3NH^+]}{[(CH_3)_3N]})](/tpl/images/0576/5686/d931d.png)
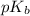 = negative logarithm of base dissociation constant of trimethylamine = 4.19
= negative logarithm of base dissociation constant of trimethylamine = 4.19![[(CH_3)_3NH^+]=\frac{0.0015}{0.0275}](/tpl/images/0576/5686/9dc68.png)
![[(CH_3)_3NH]=\frac{0.0005}{0.0275}](/tpl/images/0576/5686/ea9b4.png)