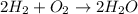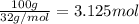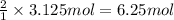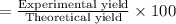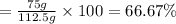Chemistry, 01.04.2020 21:45 mariarodriguezout9cj
Consider the chemical equation for the production of water: 2 H2+O2→2 H2O. If 100 grams of oxygen gas are used, what would the percent yield be if 75 g of H2O was produced? Show your work.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 14:00
Displacement is the slope of a velocity vs. time graph a. true b. false
Answers: 1

Chemistry, 22.06.2019 18:30
The table lists the lattice energies of some compounds.compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf.the lattice energy increases as the cations get larger, as shown by lif and licl.the lattice energy decreases as cations get smaller, as shown by nacl and naf.the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3

Chemistry, 22.06.2019 20:30
Identify the correct mole ratio for each substance. sodium chloride (nacl) na: cl = 1: ammonium nitrate (nhno) h: o = 4:
Answers: 1
You know the right answer?
Consider the chemical equation for the production of water: 2 H2+O2→2 H2O. If 100 grams of oxygen ga...
Questions



Geography, 28.09.2020 22:01

History, 28.09.2020 22:01

English, 28.09.2020 22:01

Biology, 28.09.2020 22:01

Chemistry, 28.09.2020 22:01

Mathematics, 28.09.2020 22:01


Mathematics, 28.09.2020 22:01



Mathematics, 28.09.2020 22:01

Mathematics, 28.09.2020 22:01

Mathematics, 28.09.2020 22:01

Mathematics, 28.09.2020 22:01


Mathematics, 28.09.2020 22:01