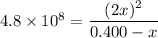H2(g) + Br2(l) ⇄ 2HBr(g) Kc = 4.8 × 108
Assume initial conditions of 0.400 M H2(g) and e...

Chemistry, 01.04.2020 20:16 CHRONICxDJ
H2(g) + Br2(l) ⇄ 2HBr(g) Kc = 4.8 × 108
Assume initial conditions of 0.400 M H2(g) and excess Br2(l). What is the equilibrium concentration of H2(g)?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 22.06.2019 15:30
The reactions of photosynthesis occur in the of plant cell? a.mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1

Chemistry, 22.06.2019 23:00
What is the solubility-product constant of barium sulfate, baso4, if a saturated solution is 1.03 ´ 10-5 m?
Answers: 3

Chemistry, 23.06.2019 00:00
(04.05 hc) analyze the given diagram of the carbon cycle below. part 1: which compound does c represent? part 2: name a process that could release this compound into the air. part 3: explain how the elements that form it are conserved during the carbon cycle. use complete sentences to explain your answer. justify how this compound was created from a recycling of carbon in the carbon cycle. use complete sentences to explain your answer.
Answers: 3
You know the right answer?
Questions

Mathematics, 25.09.2021 18:10


Law, 25.09.2021 18:10

English, 25.09.2021 18:10

Mathematics, 25.09.2021 18:10

Mathematics, 25.09.2021 18:10






Mathematics, 25.09.2021 18:10


Computers and Technology, 25.09.2021 18:10

Mathematics, 25.09.2021 18:10

Physics, 25.09.2021 18:10

Mathematics, 25.09.2021 18:10

Law, 25.09.2021 18:10

Mathematics, 25.09.2021 18:10

Social Studies, 25.09.2021 18:10

![k_c=\dfrac{[HBr(g)]^2}{[H_2]}=4.8\times 10^8M](/tpl/images/0576/0517/a3b30.png)