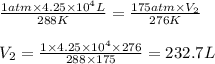Chemistry, 01.04.2020 16:58 jjjjjjgegi3088
An airtight container with a volume of 4.25 x 10^4L, an internal pressure of 1.00 atm, and an internal temperature of 15.00 *C is washed off the deck of a ship and sinks to a depth where the pressure is 175 atm and the temperature is 3.00 *C. What will the volume of the gas inside be when the container breaks under the pressure at this depth?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:20
Which statement accurately describes the relationship between air pressure, air density, or altitude? as altitude increases, pressure increases.as altitude increases, air density increases.air pressure and density are lowest at sea level.denser air exerts more pressure than less dense air.
Answers: 2

Chemistry, 22.06.2019 00:30
13. calculate the initial concentration (before precipitation) of carbonate ions after the addition of each 0.05 ml of solution b to the 1.00 l beaker of solution a. divide the work among group members and write the answers in the table in model 3. assume the volume change as solution b is added is negligible. 14. notice the initial concentrations of zn2+ - and cu2+ in the table in model 3. a. explain how these were obtained from the data in model 2. b. as solution b is added and precipitates form, do these initial concentrations change? 15. use the data in model 2 to indicate the presence of precipitate (either znco3 or cuco3) after each 0.05 ml addition of solution b in model 3. 16. use the initial concentrations of carbonate ions and zinc ions to calculate the reaction quotient, qsp for the zinc carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3. 17. use the initial concentrations of carbonate ion and copper(ii) ions to calculate the qsp for the copper(ii) carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3.
Answers: 3

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 1

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1
You know the right answer?
An airtight container with a volume of 4.25 x 10^4L, an internal pressure of 1.00 atm, and an intern...
Questions

Mathematics, 09.06.2020 05:57

Mathematics, 09.06.2020 05:57

Arts, 09.06.2020 05:57

Mathematics, 09.06.2020 05:57

Biology, 09.06.2020 05:57

Mathematics, 09.06.2020 05:57

Mathematics, 09.06.2020 05:57

Social Studies, 09.06.2020 05:57

English, 09.06.2020 05:57


Mathematics, 09.06.2020 05:57

Mathematics, 09.06.2020 05:57



History, 09.06.2020 05:57


History, 09.06.2020 05:57



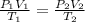
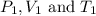 are the initial pressure, volume and temperature of the gas
are the initial pressure, volume and temperature of the gas 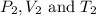 are the final pressure, volume and temperature of the gas
are the final pressure, volume and temperature of the gas![P_1=1.00atm\\V_1=4.25\times 10^4L\\T_1=15^oC=[15+273]K=288K\\P_2=175atm\\V_2=?\\T_2=3^oC=[3+273]K=276K](/tpl/images/0575/4914/4be35.png)