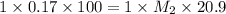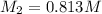Chemistry, 31.03.2020 01:05 bjpvrpow74wq
To standardize a solution of NaOH before using it in a titration of an unknown acid, you dissolve 3.56 grams of potassium hydrogen phthalate (KHP) into 100 mL of H2O. You then titrate the KHP with your sodium hydroxide solution and reach the endpoint after adding 20.9 mL of NaOH. What is the molarity of your sodium hydroxide solution? (Molecular Mass of KHP = 204.22 g/mol)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:20
Use the gizmo to find the concentration of the mystery ch3cooh. use the titrant and indicator shown below perform the titration. what is the titrant volume? titrant analyte indicator titrant volume analyte concentration naoh ch3cooh phenophthalein select one: a. 20.0 ml b. 27.0 ml c. 30.0 ml d. 24.0 ml
Answers: 2

Chemistry, 22.06.2019 06:00
24. a sports ball is inflated to an internal pressure of 1.85 atm at room temperature (25 °c). if the ball is then played with outside where the temperature is 7.5 °c, what will be the new pressure of the ball? assume the ball does not change in volume nor does any air leak from the ball a) 0.555 atm b) 1.74 atm c) 1.85 atm d) 1.97 atm
Answers: 2

Chemistry, 22.06.2019 15:40
Use the periodic table to complete this equation that represents nuclear fission processesun - ba c 3 n
Answers: 2

Chemistry, 22.06.2019 19:10
Δu of , in kj/kg, as it isto k, (a)as a of , (b) at , (c) at .
Answers: 2
You know the right answer?
To standardize a solution of NaOH before using it in a titration of an unknown acid, you dissolve 3....
Questions

Mathematics, 10.10.2019 03:40

Physics, 10.10.2019 03:40





Mathematics, 10.10.2019 03:40




Chemistry, 10.10.2019 03:40





History, 10.10.2019 03:40

Mathematics, 10.10.2019 03:40

Mathematics, 10.10.2019 03:40

English, 10.10.2019 03:40

Social Studies, 10.10.2019 03:40

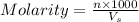
 = volume of solution in ml = 100 ml
= volume of solution in ml = 100 ml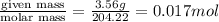
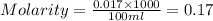
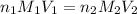
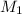 = molarity of KHP solution = 0.17 M
= molarity of KHP solution = 0.17 M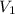 = volume of KHP solution = 100 ml
= volume of KHP solution = 100 ml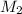 = molarity of NaOH solution = ?
= molarity of NaOH solution = ?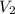 = volume of NaOH solution = 20.9 ml
= volume of NaOH solution = 20.9 ml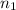 = valency of KHP = 1
= valency of KHP = 1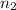 = valency of NaOH = 1
= valency of NaOH = 1