
Chemistry, 31.03.2020 00:14 aubreyfoster
Express the rate of the reaction in terms of the change in concentration of each of the reactants and products.
a. rate=12Δ[HBr]Δt=−Δ[H2]Δt=−Δ[Br2]Δtr ate=12Δ[HBr]Δt=−Δ[H2]Δt=−Δ[Br2]Δt
b. rate=−Δ[HBr]Δt=12Δ[H2]Δt=12Δ[Br2]Δt rate=−Δ[HBr]Δt=12Δ[H2]Δt=12Δ[Br2]Δt
c. rate=−12Δ[HBr]Δt=Δ[H2]Δt=Δ[Br2]Δtra te=−12Δ[HBr]Δt=Δ[H2]Δt=Δ[Br2]Δt
d. rate=Δ[HBr]Δt=−12Δ[H2]Δt=−12Δ[Br2]Δ t

Answers: 3
Another question on Chemistry



Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 2
You know the right answer?
Express the rate of the reaction in terms of the change in concentration of each of the reactants an...
Questions



Biology, 10.10.2021 01:00

Mathematics, 10.10.2021 01:00



History, 10.10.2021 01:00


Mathematics, 10.10.2021 01:00


Mathematics, 10.10.2021 01:00

Health, 10.10.2021 01:00

Business, 10.10.2021 01:00




English, 10.10.2021 01:00



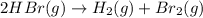
![rate=-\frac{1}{2} \frac{\Delta [HBr]}{\Delta t}=\frac{\Delta [Br_2]}{\Delta t} =\frac{\Delta [H_2]}{\Delta t}](/tpl/images/0572/0368/fc117.png)


