Chemistry, 30.03.2020 23:46 abieber4328
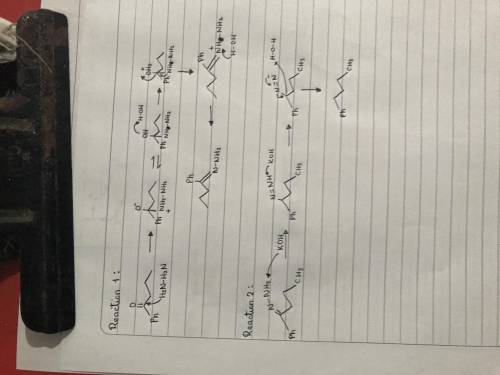
The Wolff-Kishner reaction involves the reaction of an aldehyde/ketone with hydrazine in the presence of KOH. The process is useful for converting an aldehyde or ketone into an alkane. The reaction involves formation of a hydrazone, followed by base-catalyzed double-bond migration, loss of N2 gas to give a carbanion, and protonation to give the alkane. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3

Chemistry, 22.06.2019 17:30
Air can be considered a mixture. which statement does not explain why?
Answers: 1

Chemistry, 22.06.2019 20:30
Which of the following is not true about the atomic model of substances?
Answers: 1

Chemistry, 22.06.2019 21:30
Achemical reaction is done in the setup shown, resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 1
You know the right answer?
The Wolff-Kishner reaction involves the reaction of an aldehyde/ketone with hydrazine in the presenc...
Questions




Mathematics, 17.02.2021 21:20


Biology, 17.02.2021 21:20





History, 17.02.2021 21:20

Mathematics, 17.02.2021 21:20

Mathematics, 17.02.2021 21:20

Mathematics, 17.02.2021 21:20

Mathematics, 17.02.2021 21:20


Mathematics, 17.02.2021 21:20

Biology, 17.02.2021 21:20


Physics, 17.02.2021 21:20