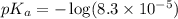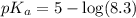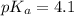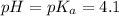The half‑equivalence point of a titration occurs half way to the equivalence point, where half of the analyte has reacted to form its conjugate, and the other half still remains unreacted. If 0.440 moles of a monoprotic weak acid ( K a = 8.3 × 10 − 5 ) is titrated with NaOH , what is the pH of the solution at the half‑equivalence point?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:30
Given that the molar mass of nano3 is 85.00 g/mol, what mass of nano3 is needed to make 4.50 l of a 1.50 m nano3solution? use .6.75 g18.9 g255 g574 g
Answers: 1

Chemistry, 22.06.2019 00:10
According to the diagram; a) identify the anode of the cell and write the half-reaction that occurs there b) write the overall equation for the reaction that occurs as the cell operates c) calculate the value of the standard cell potential ,e cell. d)write the shorthand notation of the cell above e)indicate the flow of the electrons on the diagram
Answers: 3

Chemistry, 22.06.2019 15:00
What is the most important factor in determining climates.
Answers: 1

Chemistry, 22.06.2019 20:00
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2
You know the right answer?
The half‑equivalence point of a titration occurs half way to the equivalence point, where half of th...
Questions

Medicine, 27.06.2021 16:50


Mathematics, 27.06.2021 16:50

Social Studies, 27.06.2021 16:50

Mathematics, 27.06.2021 16:50

Physics, 27.06.2021 16:50

Mathematics, 27.06.2021 16:50


Mathematics, 27.06.2021 16:50

Mathematics, 27.06.2021 16:50


Physics, 27.06.2021 16:50


Social Studies, 27.06.2021 16:50

Mathematics, 27.06.2021 16:50


Mathematics, 27.06.2021 16:50

Social Studies, 27.06.2021 16:50


Mathematics, 27.06.2021 17:00

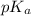 of weak acid.
of weak acid.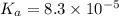
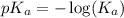
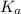 in this expression, we get:
in this expression, we get: