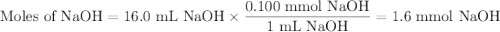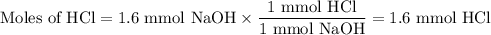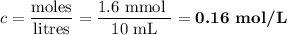Chemistry, 30.03.2020 22:52 gghkooo1987
Base your answer on the information below and on your knowledge of chemistry.
A NaOH(aq) solution with a pH value of 13 is used to determine the molarity of an HCl(aq) solution. A 10.0-mL sample of the HCl(aq) is exactly neutralized by 16.0 mL of 0.100 M NaOH(aq). During this laboratory activity, appropriate safety equipment was used and safety procedures were followed.
Determine the pH value of a solution that has a H+(aq) ion concentration 10 times greater than the original NaOH(aq) solution.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:30
What is the relation between concentration of reactants and the rate of chemical reaction?
Answers: 1


Chemistry, 22.06.2019 21:00
What type of radiation is lead emitting in the following equation? alpha particles beta particles gamma rays
Answers: 3

Chemistry, 22.06.2019 23:10
Using the periodic table, complete the following. element: hydrogen symbol: h₂ molecular weight: g mass of one mole: g/mol
Answers: 3
You know the right answer?
Base your answer on the information below and on your knowledge of chemistry.
A NaOH(aq)...
A NaOH(aq)...
Questions


Mathematics, 11.01.2021 05:40

Mathematics, 11.01.2021 05:40

Business, 11.01.2021 05:40





Mathematics, 11.01.2021 05:50


Biology, 11.01.2021 05:50


Mathematics, 11.01.2021 05:50




Biology, 11.01.2021 05:50

Mathematics, 11.01.2021 05:50

Mathematics, 11.01.2021 05:50

Biology, 11.01.2021 05:50