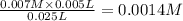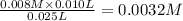A student working in the laboratory prepared the following reactants: 5 mL of 0.007M Cd2+(aq) 10 mL of 0.008M SCN-(aq) 10 mL of 0.5M HNO3(aq) These reagents were mixed and allowed to stand for 10 minutes. The concentration of Cd(SCN)+ in the resulting equilibrium mixture is found to be 5 x 10−4M. Calculate the initial concentration of Cd2+(aq).

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:50
Choose all that apply. when creating a graph, you should: determine the x- and y- variables label the scale on the x- and y- axes plot the data points draw a line of best fit to represent the data trend
Answers: 1

Chemistry, 21.06.2019 21:00
Which of the following compounds does not contain molecules? question 2 options: co2 h2 nacl h2o
Answers: 1

Chemistry, 22.06.2019 21:00
As we move from left to right across the periodic table, what is the general trend? a) atomic radii increase. b) electronegavitiy decreases. c) nuclear shielding increases. d) metallic character decreases.
Answers: 1

Chemistry, 22.06.2019 23:30
Substance a is a nonpolar liquid and has only dispersion forces among its constituent particles. substance b is also a nonpolar liquid and has about the same magnitude of dispersion forces among its constituent particles. when substance a and b are combined, they spontaneously mix.
Answers: 1
You know the right answer?
A student working in the laboratory prepared the following reactants: 5 mL of 0.007M Cd2+(aq) 10 mL...
Questions



History, 27.05.2021 04:10

Mathematics, 27.05.2021 04:10


Biology, 27.05.2021 04:20




Mathematics, 27.05.2021 04:20

English, 27.05.2021 04:20

Mathematics, 27.05.2021 04:20

Mathematics, 27.05.2021 04:20




English, 27.05.2021 04:20


Mathematics, 27.05.2021 04:20

Chemistry, 27.05.2021 04:20

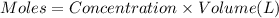
![[Cd^{2+}]=0.007M](/tpl/images/0571/8149/5e85a.png)
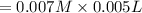
![[SCN^{-}]=0.008 M](/tpl/images/0571/8149/33083.png)
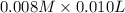
![[HNO_3]=0.5 M](/tpl/images/0571/8149/6e078.png)