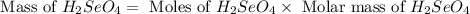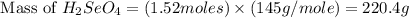Chemistry, 30.03.2020 22:32 savannahvargas512
Determine the limiting reactant in a mixture containing 139 g of Se, 431 g of Cl2, and 110 g of H2O. Calculate the maximum mass (in grams) of selenic acid, H2SeO4, that can be produced in the reaction.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 23.06.2019 01:00
The time that is taken by neptune once around the sun is called
Answers: 1

Chemistry, 23.06.2019 13:30
The two isotopes of chlorine are 3517cl and 3717cl. which isotope is the most abundant?
Answers: 1

Chemistry, 23.06.2019 14:00
Which statement describes the arrhenius interpretation of acids and bases?
Answers: 1
You know the right answer?
Determine the limiting reactant in a mixture containing 139 g of Se, 431 g of Cl2, and 110 g of H2O....
Questions


Mathematics, 16.12.2020 02:40

English, 16.12.2020 02:40

Mathematics, 16.12.2020 02:40

Physics, 16.12.2020 02:40





Mathematics, 16.12.2020 02:40

History, 16.12.2020 02:40

Mathematics, 16.12.2020 02:40

Mathematics, 16.12.2020 02:40


Mathematics, 16.12.2020 02:40

Mathematics, 16.12.2020 02:40

Mathematics, 16.12.2020 02:40


Chemistry, 16.12.2020 02:40

Mathematics, 16.12.2020 02:40

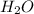
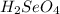 produced is, 220.4 grams.
produced is, 220.4 grams.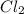 = 431 g
= 431 g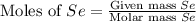
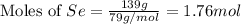
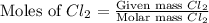
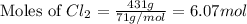
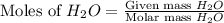
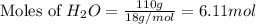
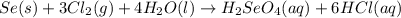
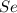
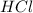 react with
react with 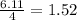 moles of
moles of 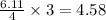 moles of
moles of 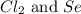 are excess reagent because the given moles are greater than the required moles and
are excess reagent because the given moles are greater than the required moles and