
Chemistry, 30.03.2020 21:01 gungamer720
G Identify limiting reactants (mole ratio method). Close Problem Identify the limiting reactant in the reaction of bromine and chlorine to form BrCl, if 29.7 g of Br2 and 11.2 g of Cl2 are combined. Determine the amount (in grams) of excess reactant that remains after the reaction is complete.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 20:20
Concerning the 10.0 ml of 0.50 m nacl to 100 ml of solution: when a solution is diluted, does it change the number of moles dissolved?
Answers: 3

Chemistry, 21.06.2019 21:00
Consider the nuclear equation below. 239 > x + 4 he 94 2 what is x? 1.235 cm 96 2.243 u 92 3.235 u 92 4.243 cm 96
Answers: 2

Chemistry, 21.06.2019 21:30
The reaction q+r2=r2q is found to be first order in r2 and
Answers: 1

Chemistry, 22.06.2019 04:00
The image shows a process that releases nuclear energy which statement best identifies the process shown the process must be fusion because energy is released the process must be fusion because of have your nucleus formed a smaller nuclei the process must be fission because a large nucleus breaks into smaller nuclei the process must be fission because neutrons are formed
Answers: 1
You know the right answer?
G Identify limiting reactants (mole ratio method). Close Problem Identify the limiting reactant in t...
Questions


Advanced Placement (AP), 04.08.2019 01:00


Mathematics, 04.08.2019 01:00

Biology, 04.08.2019 01:00


History, 04.08.2019 01:00

History, 04.08.2019 01:00

English, 04.08.2019 01:00



Biology, 04.08.2019 01:00


English, 04.08.2019 01:00


Chemistry, 04.08.2019 01:00

Chemistry, 04.08.2019 01:00



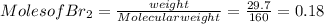
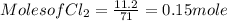
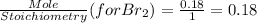
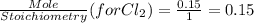
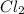 is a limiting reactant and Br₂ is excess reactant.
is a limiting reactant and Br₂ is excess reactant.

