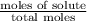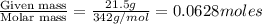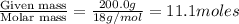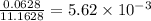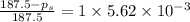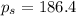Chemistry, 30.03.2020 21:00 shelatzcreed
Calculate the vapor pressure of water above a solution prepared by adding 21.5 g of lactose (C12H22O11) to 200.0 g of water at 338 K. (Vapor-pressure of water at 338 K 187.5 torr.)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 14:30
Need ! asap will mark 10 pts using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers
Answers: 1

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 3

Chemistry, 22.06.2019 20:20
Which symbol can be used to indicate the pressure at which a chemical reaction is carried out? 25°c 2 atm pa
Answers: 2

Chemistry, 23.06.2019 00:00
#7 how does the structure of amino acids allow them to form a polypeptide? each amino acid has an amino group and a carboxyl group. each amino acid has a hydrogen atom and a carboxyl group. each amino acid has a carboxyl group and an r group. each amino acid has an r group and a hydrogen atom.
Answers: 1
You know the right answer?
Calculate the vapor pressure of water above a solution prepared by adding 21.5 g of lactose (C12H22O...
Questions

Mathematics, 08.10.2019 06:30

Biology, 08.10.2019 06:30



History, 08.10.2019 06:30

History, 08.10.2019 06:30

Spanish, 08.10.2019 06:30

Mathematics, 08.10.2019 06:30


History, 08.10.2019 06:30


Mathematics, 08.10.2019 06:30



History, 08.10.2019 06:30

Mathematics, 08.10.2019 06:30

Health, 08.10.2019 06:30


Spanish, 08.10.2019 06:30

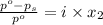
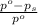 = relative lowering in vapor pressure
= relative lowering in vapor pressure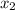 = mole fraction of solute =
= mole fraction of solute =