
Chemistry, 30.03.2020 21:06 annagwhiteou0hrh
Calculate the standard entropy change for the reaction: 2 hydrogen sulfide(g) plus sulfur dioxide (g) goes to form 3 sulfur(s) plus 2 water(g). Given S(s) = 31.88 J/mol. Look up the other values in the text appendix. No units required. Just the numerical answer

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:50
Write the overall equation for the reaction for lithium battery
Answers: 2

Chemistry, 22.06.2019 20:00
The volume of a single vanadium atom is 9.29×10-24 cm3. what is the volume of a vanadium atom in microliters?
Answers: 3

Chemistry, 23.06.2019 00:00
This statement about matter and its behavior is best classified as a
Answers: 1

Chemistry, 23.06.2019 00:30
Five different substances are given to you to be dissolved in water. which substances are most likely to undergo dissolution in water? check all that apply. view available hint(s) check all that apply. sodium fluoride, naf octane, c8h18 propanol, ch3ch2ch2oh potassium iodide, ki benzene, c6h6
Answers: 1
You know the right answer?
Calculate the standard entropy change for the reaction: 2 hydrogen sulfide(g) plus sulfur dioxide (g...
Questions

Mathematics, 04.02.2021 23:30

Mathematics, 04.02.2021 23:30

Advanced Placement (AP), 04.02.2021 23:30

Mathematics, 04.02.2021 23:30


Mathematics, 04.02.2021 23:30




Mathematics, 04.02.2021 23:30






Spanish, 04.02.2021 23:30


Chemistry, 04.02.2021 23:30

Mathematics, 04.02.2021 23:30

![\Delta S^o_{rxn}=\sum [n\times \Delta S^o_{(product)}]-\sum [n\times \Delta S^o_{(reactant)}]](/tpl/images/0571/2033/52737.png)
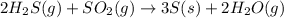
![\Delta S^o_{rxn}=[(3\times \Delta S^o_{(S(s))})+(2\times \Delta S^o_{(H_2O(g))})]-[(2\times \Delta S^o_{(H_2S(g))})+(1\times \Delta S^o_{(SO_2(g))})]](/tpl/images/0571/2033/492b6.png)
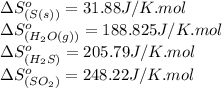
![\Delta S^o_{rxn}=[(3\times (31.88))+(2\times (188.825))]-[(2\times (205.79))+(1\times (248.22))]\\\\\Delta S^o_{rxn}=-186.51J/K](/tpl/images/0571/2033/7cc5d.png)


