Chemistry, 30.03.2020 20:35 esanchez2002fcb
Dry ice is solid carbon dioxide. A 1.65−g sample of dry ice is placed in an evacuated 3.93−L vessel at 23.0°C. Calculate the pressure inside the vessel after all the dry ice has been converted to CO2 gas.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:40
Which characteristic of water it form droplets? a. low specific heat b. nonpolar structure c. high surface tension d. ability to dissolve substances
Answers: 1

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 07:00
Which atom or ion is the largest? a. k b. k+ c. ca d. ca2+ e. li
Answers: 1

Chemistry, 22.06.2019 07:30
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 3
You know the right answer?
Dry ice is solid carbon dioxide. A 1.65−g sample of dry ice is placed in an evacuated 3.93−L vessel...
Questions



Mathematics, 30.07.2021 20:20

Computers and Technology, 30.07.2021 20:20







Mathematics, 30.07.2021 20:20

Mathematics, 30.07.2021 20:20







Health, 30.07.2021 20:20

Mathematics, 30.07.2021 20:20

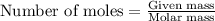
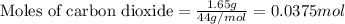
![23^oC=[23+273]=296K](/tpl/images/0571/0457/eb892.png)