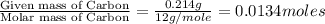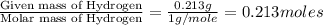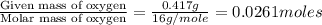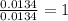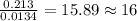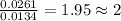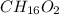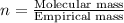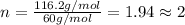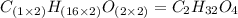Chemistry, 30.03.2020 21:13 robertjoy19
The smell of dirty gym socks is caused by the compound caproic acid(contains H, C, O). Combustion of 0.844 g of caproic acid produced 0.784 g of H2O and 1.92 g of CO2. If the molar mass of caproic acid is 116.2 g/mol, what is the molecular formula of caproic acid? (MM C = 12g/mol, H = 1g/mol, O = 16 g/mol)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 14:30
Which of the following is not one of the steps in the scientific method a. hypothesize b. summarize c. analyze d. familiarize
Answers: 3

Chemistry, 22.06.2019 22:30
Which process describes vaporization that takes place below the surface of a liquid? condensation melting boiling evaporation
Answers: 1

Chemistry, 22.06.2019 23:00
The data below were determined for the reaction shown below. s2o82– + 3i – (aq) → 2so42– + i3– expt. # [s2o82–] (m) [i –] (m) initial rate 1 0.038 0.060 1.4 × 10 – 5 m/s 2 0.076 0.060 2.8 × 10 – 5 m/s 3 0.076 0.030 1.4 × 10 – 5 m/s the rate law for this reaction must be:
Answers: 1

Chemistry, 23.06.2019 00:00
How many peaks will be present in a mass spectrum for brcl?
Answers: 1
You know the right answer?
The smell of dirty gym socks is caused by the compound caproic acid(contains H, C, O). Combustion of...
Questions


Biology, 14.02.2022 07:20

English, 14.02.2022 07:20


World Languages, 14.02.2022 07:20



History, 14.02.2022 07:20

Mathematics, 14.02.2022 07:20

Geography, 14.02.2022 07:20





English, 14.02.2022 07:20


Mathematics, 14.02.2022 07:20

Mathematics, 14.02.2022 07:20


Mathematics, 14.02.2022 07:20

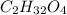
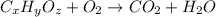
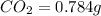
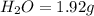
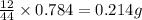 of carbon will be contained.
of carbon will be contained.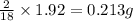 of hydrogen will be contained.
of hydrogen will be contained.