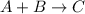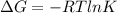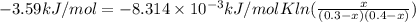Chemistry, 30.03.2020 20:50 Elepeodowke
Has a standard free‑energy change of − 3.59 kJ / mol at 25 °C. What are the concentrations of A , B , and C at equilibrium if, at the beginning of the reaction, their concentrations are 0.30 M, 0.40 M, and 0 M, respectively?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Consider the nuclear equation below. 239 > x + 4 he 94 2 what is x? 1.235 cm 96 2.243 u 92 3.235 u 92 4.243 cm 96
Answers: 2

Chemistry, 22.06.2019 00:30
You have 125g of a certain seasoning and are told that it contains 76.0 g of salt what is the percentage of salt by mass in this seasoning
Answers: 1

Chemistry, 22.06.2019 10:50
A100 kmol/h stream that is 97 mole% carbon tetrachloride (ccl4) and 3% carbon disulfide (cs2) is to be recovered from the bottom of a distillation column. the feed to the column is 16 mole% cs2 and 84% ccl4, and 2% of the ccl4 entering the column is contained in the overhead stream leaving the top of the column. calculate the mass and mole fractions of ccl4 in the overhead stream, and determine the molar flow rates of ccl4 and cs2 in the overhead and feed streams. 12. mw_ccla- 153.82; mw_cs2-76.14.
Answers: 3

Chemistry, 22.06.2019 11:00
The diagram below shows the different phase transitions that occur in matter. which arrow represents the transition in which dew is formed?
Answers: 1
You know the right answer?
Has a standard free‑energy change of − 3.59 kJ / mol at 25 °C. What are the concentrations of A , B...
Questions

Social Studies, 21.09.2019 09:10

Chemistry, 21.09.2019 09:10





English, 21.09.2019 09:10



English, 21.09.2019 09:10


Social Studies, 21.09.2019 09:10

Business, 21.09.2019 09:10

Health, 21.09.2019 09:10

Mathematics, 21.09.2019 09:10



Mathematics, 21.09.2019 09:10

Chemistry, 21.09.2019 09:10

Social Studies, 21.09.2019 09:10