

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Reaction rate depends on how many molecules are coming into contact with each other with enough energy to react. increasing the temperature of the reactants will increase -
Answers: 3


Chemistry, 23.06.2019 07:00
Under what conditions will a gas be most likely to exhibit the ideal gas properties predicted by the ideal gas law? 1)high pressures and high temperature, because particles are forced closer together with higher kinetic energy, so intermolecular forces of attraction are weaker 2)high pressure and low temperature, because particles are forced closer together and moving slower, so the volume of the particles is less significant 3) low pressure and high temperature, because particles are spread farther apart and moving faster, so the intermolecular forces of attraction are weaker 4)low pressure and low temperature, because particles are spread farther apart with lower kinetic energy, so the volume of the particles is less significant
Answers: 2

Chemistry, 23.06.2019 10:50
Which compound should undergo substitution of the bromine by phenolate anion? draw the structure of the organic product?
Answers: 1
You know the right answer?
A solution containing CaCl2 is mixed with a solution of Li2C2O4 to form a solution that is 3.5 × 10-...
Questions


Mathematics, 20.09.2020 15:01





Physics, 20.09.2020 15:01





Mathematics, 20.09.2020 15:01

English, 20.09.2020 15:01


Mathematics, 20.09.2020 15:01

English, 20.09.2020 15:01



Mathematics, 20.09.2020 15:01

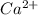 =
= 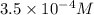
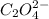 =
= 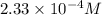
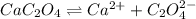
![K_{sp}=[Ca^{2+}][C_2O_4^{2-}]=2.3\times 10^{-9}](/tpl/images/0570/7450/df299.png)
![Q_{sp}=[Ca^{2+}][C_2O_4^{2-}]](/tpl/images/0570/7450/af00c.png)
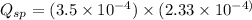
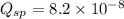
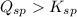 that means a white solid precipitate of calcium oxalate will be formed when the solutions are mixed.
that means a white solid precipitate of calcium oxalate will be formed when the solutions are mixed.

