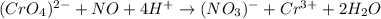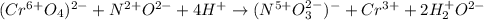Chemistry, 30.03.2020 17:02 naomicervero
CrO42- + NO+ 4H+NO3- + Cr3++ 2H2O In the above redox reaction, use oxidation numbers to identify the element oxidized, the element reduced, the oxidizing agent and the reducing agent. name of the element oxidized: name of the element reduced: formula of the oxidizing agent: formula of the reducing agent:

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 19:30
Water molecules have a strong attraction to each other because of hydrogen bonding, allowing water to move against gravity up a plant's stem through capillary action. true false
Answers: 2

Chemistry, 21.06.2019 21:10
Identify one disadvantage to each of the following models of electron configuration: dot structures arrow and line diagrams written electron configurations type in your answer below.
Answers: 1

Chemistry, 22.06.2019 10:30
Which describes fat? a: a carbohydrate that produces energy b: a nucleic acid that directs cell function c: a lipid that stores energy d: a protein that speeds up a chemical reaction
Answers: 1

Chemistry, 22.06.2019 18:30
How many moles of bromine are needed to produce 3.23 moles of potassium bromide
Answers: 1
You know the right answer?
CrO42- + NO+ 4H+NO3- + Cr3++ 2H2O In the above redox reaction, use oxidation numbers to identify the...
Questions


Chemistry, 02.04.2020 20:01

Mathematics, 02.04.2020 20:01








History, 02.04.2020 20:02


Mathematics, 02.04.2020 20:02



Computers and Technology, 02.04.2020 20:02

Business, 02.04.2020 20:02



Mathematics, 02.04.2020 20:02