
Chemistry, 30.03.2020 04:33 smelcher3900
According to the equation below, how many moles of Ca(OH)2 are required to react with 1.36 mol H3PO4 to produce Ca3(PO4)2? 3Ca(OH)2+2H3PO4⟶Ca3(PO4)2+6H2O

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:30
The is a particle with one unit of positive charge a. proton b. positron c. electron d. nucleus awnser quick it is a important science test!
Answers: 2

Chemistry, 22.06.2019 03:00
Zoe is investigating the composition of substance a, an unknown substance. using chemical processes, she analyzes substance a and determines it is composed of sodium, oxygen, and hydrogen atoms in a ratio of 1 : 1 : 1. what is substance a? a. a compound b. an element c. a heterogeneous mixture d. a homogeneous mixture
Answers: 1

Chemistry, 22.06.2019 09:30
Why do cells appear different in distilled water than they do in 10% salt water?
Answers: 2

Chemistry, 22.06.2019 10:00
3. how much energy in joules is required to evaporate .0005 kg of liquid ammonia to vapor at the same temperature? 4. how much energy ( in megajoules ) is given up by .75 kg of water at 0c when it freezes to form ice at 0c? 5. explain how heat works between and at critical temperatures?
Answers: 2
You know the right answer?
According to the equation below, how many moles of Ca(OH)2 are required to react with 1.36 mol H3PO4...
Questions

English, 09.11.2021 06:10


History, 09.11.2021 06:20


Health, 09.11.2021 06:20

Mathematics, 09.11.2021 06:20

Mathematics, 09.11.2021 06:20

English, 09.11.2021 06:20







Mathematics, 09.11.2021 06:30


Biology, 09.11.2021 06:30

Mathematics, 09.11.2021 06:30

Business, 09.11.2021 06:30

Mathematics, 09.11.2021 06:30

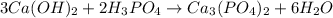
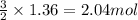 of calcium hydroxide
of calcium hydroxide

