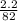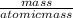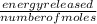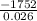A calorimeter contains 30.0 mL of water at 12.5 ∘C . When 2.20 g of X (a substance with a molar mass of 82.0 g/mol ) is added, it dissolves via the reaction X(s)+H2O(l)→X(aq) and the temperature of the solution increases to 25.5 ∘C . Calculate the enthalpy change, ΔH, for this reaction per mole of X. Assume that the specific heat of the resulting solution is equal to that of water [4.18 J/(g⋅∘C)], that density of water is 1.00 g/mL, and that no heat is lost to the calorimeter itself, nor to the surroundings

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:00
What forms when chemical reactions combine pollution with sunlight?
Answers: 1


Chemistry, 23.06.2019 04:00
What changes occur in the reaction indicated by the equation? check all that apply. the hydrogen nucleus loses protons. the oxygen nucleus gains protons. the bond in h2 is broken, and new bonds are formed between hydrogen and oxygen atoms. each electron associated with a hydrogen atom is shared with an oxygen atom.
Answers: 3

Chemistry, 23.06.2019 04:10
What does the field of thermodynamics relate to a-changes in nuclear reactions b- changes in energy in systems c changes in molecular structure d changes in atomic properties
Answers: 1
You know the right answer?
A calorimeter contains 30.0 mL of water at 12.5 ∘C . When 2.20 g of X (a substance with a molar mass...
Questions


Mathematics, 29.09.2020 21:01


Business, 29.09.2020 21:01

Biology, 29.09.2020 21:01


Mathematics, 29.09.2020 21:01


English, 29.09.2020 21:01

Mathematics, 29.09.2020 21:01






History, 29.09.2020 21:01