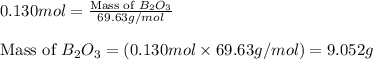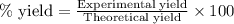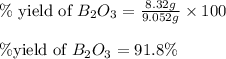Chemistry, 29.03.2020 21:45 demarpratt1270
Find percent yield:
The mass of B2O3 produced by the reaction of 4.00 g of B5H9, and 10.00 g of
O2 is 8.32 g. What is the percent yield?
2 B3H9 + 12 O2 => 5 B2O3 +9 H2O
87.2
92.8
91.8
75.5
74.5

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:00
In the 1800s, one of the statements in john dalton's atomic theory was that atoms are indivisible. later experimental evidence led to the discovery of subatomic particles such as neutrons, electrons, and protons. what happened to the indivisible atom part of dalton's atomic theory, and why?
Answers: 3

Chemistry, 22.06.2019 06:00
If you burn 10 kilograms of wood in a fire (combustion) what is the weight of the products after the fire has finished burning the wood?
Answers: 3

Chemistry, 22.06.2019 22:30
Draw the aromatic compound toluene (methylbenzene). show all hydrogen atoms, including those on the ring.
Answers: 1

Chemistry, 23.06.2019 03:50
What is the equation fort the alkaline zinc/manganese dioxide cell. a) anode b)cathode c)overall equations.
Answers: 2
You know the right answer?
Find percent yield:
The mass of B2O3 produced by the reaction of 4.00 g of B5H9, and 10....
The mass of B2O3 produced by the reaction of 4.00 g of B5H9, and 10....
Questions

Mathematics, 14.05.2021 04:10

Computers and Technology, 14.05.2021 04:10

Mathematics, 14.05.2021 04:10

Arts, 14.05.2021 04:10


Mathematics, 14.05.2021 04:10




Biology, 14.05.2021 04:10

Mathematics, 14.05.2021 04:10

Mathematics, 14.05.2021 04:10


Mathematics, 14.05.2021 04:10



English, 14.05.2021 04:10


Mathematics, 14.05.2021 04:10

Biology, 14.05.2021 04:10

 .....(1)
.....(1)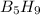 :
: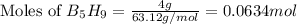
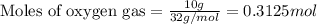
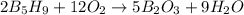
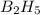
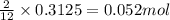 of
of 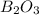
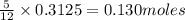 of water
of water