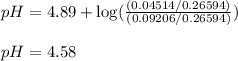Chemistry, 28.03.2020 04:02 cupcake20019peehui
An analytical chemist is titrating of a solution of propionic acid with a solution of 224.9 ml of a 0.6100M solution of propionic acid (HC2H5CO2) with a 1.1000M solution of KOH. The pKa of proionic acid 4.89.
Calculate the pH of the acid solution after the chemist has added 41.04mL of the KOH solution to it.
Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of solution added. Round your answer to decimal places.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 19:30
Which answer lists the fundamental forces in order from strongest to weakest
Answers: 1

Chemistry, 21.06.2019 22:00
Describe the particles of all three phases of matter in the root beer float. (how are the particles arranged and moving? )
Answers: 3

Chemistry, 22.06.2019 01:00
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3
You know the right answer?
An analytical chemist is titrating of a solution of propionic acid with a solution of 224.9 ml of a...
Questions




English, 03.03.2020 21:45




Physics, 03.03.2020 21:45


Computers and Technology, 03.03.2020 21:45

Mathematics, 03.03.2020 21:45




Biology, 03.03.2020 21:45


English, 03.03.2020 21:45

Mathematics, 03.03.2020 21:45


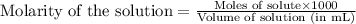 .....(1)
.....(1)
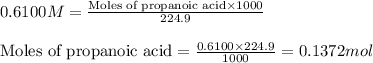
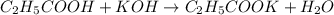
![pH=pK_a+\log(\frac{[\text{salt}]}{[acid]})](/tpl/images/0568/6689/3d096.png)
![pH=pK_a+\log(\frac{[C_2H_5COOK]}{[C_2H_5COOH]})](/tpl/images/0568/6689/3ede9.png)
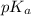 = negative logarithm of acid dissociation constant of propanoic acid = 4.89
= negative logarithm of acid dissociation constant of propanoic acid = 4.89![[C_2H_5COOK]=\frac{0.04514}{0.26594}](/tpl/images/0568/6689/11196.png)
![[C_2H_5COOH]=\frac{0.09206}{0.26594}](/tpl/images/0568/6689/86178.png)