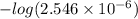Chemistry, 27.03.2020 16:43 clarkster112679
A chemist titrates 110.0 mL of a 0.7684 M methylamine (CH3NH2) sotion with 0.4469 M HNO3 solution at 25 °C. Calculate the pH at equivalence. The p K, of methylamine is 3.36 Round your answer to 2 decimal places Note for advanced students: you may assume the total volume of the solution equals the initial volume plus the volume of HNO3 solution added

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:20
Why does his teacher ask him to balance the equation by including the correct coefficient
Answers: 1

Chemistry, 22.06.2019 10:00
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 22.06.2019 18:10
Areader can tell that the meaning of “obnoxious” will include “having the quality of something” because of the .a) prefix b)pronunciation c)suffix d) word root
Answers: 3
You know the right answer?
A chemist titrates 110.0 mL of a 0.7684 M methylamine (CH3NH2) sotion with 0.4469 M HNO3 solution at...
Questions

Biology, 12.12.2019 04:31

History, 12.12.2019 04:31

History, 12.12.2019 04:31




Mathematics, 12.12.2019 04:31



English, 12.12.2019 04:31


History, 12.12.2019 04:31

Mathematics, 12.12.2019 04:31

Chemistry, 12.12.2019 04:31

Biology, 12.12.2019 04:31

Chemistry, 12.12.2019 04:31

English, 12.12.2019 04:31

Mathematics, 12.12.2019 04:31


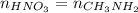
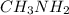 as follows.
as follows.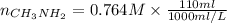
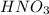 present will be calculated as follows.
present will be calculated as follows.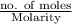
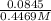
![[CH_{3}NH_{3}^{+}] = \frac{0.0845 mol}{0.2991 L}](/tpl/images/0567/3743/df2f2.png)
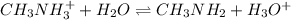
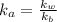
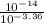
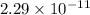
![[HNO_{3}] = \sqrt{k_{a}[CH_{3}NH_{3}^{+}]}](/tpl/images/0567/3743/a246e.png)
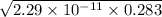
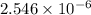
![-log [H_{3}O^{+}]](/tpl/images/0567/3743/df803.png)