
Chemistry, 27.03.2020 01:50 marsewilliams
Suppose the reaction system below has already reached equilibrium. Predict the effect that each of the following changes will have on the equilibrium position. Tell whether the equilibrium will shift to the right, will shift to the left, or will not be affected.
UO2(s) + 4 HF(g) ↔ UF4(g) + 2 H2O(g)
1. Water vapor is removed
2. UF4(g) is added
3. The reaction is done in a glass reaction vessel. HF(g) attacks and reacts with the glass
4. If the reaction is endothermic, and you want to make the equilibrium shift to the right to maximize the products, would you heat or cool the reaction vessel?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:30
A3.37-mg sample of protein was chemically digested to convert its nitrogen into ammonia and then diluted to 100.0 ml. then 10.0 ml of this solution was placed in a 50-ml volumetric flask and treated with 5 ml of phenol solution plus 2 ml of sodium hypochlorite solution. the sample was diluted to 50.0 ml, and the absorbance at 625 nm was measured in a 1.00-cm cuvette and found to be 0.486. for reference, a standard solution was prepared from 10.0 mg of nh4cl (molar mass = 53.49 grams/mole) dissolved in 1.00 l of water. then 10.0 ml of this standard was placed in a 50-ml volumetric flask, treated in the same manner as the unknown, and the absorbance found to be 0.323. finally, a reagent blank was prepared using distilled water in place of unknown, it was treated in the same manner as the unknown, and the absorbance found to be 0.076. calculate the weight percent of nitrogen in the protein.
Answers: 1


Chemistry, 22.06.2019 08:30
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 1

Chemistry, 22.06.2019 13:10
Which electron configuration represents the electrons in an atom of sodium in the ground state at stp
Answers: 1
You know the right answer?
Suppose the reaction system below has already reached equilibrium. Predict the effect that each of t...
Questions


Mathematics, 05.10.2019 15:30

Social Studies, 05.10.2019 15:30

Mathematics, 05.10.2019 15:30

Mathematics, 05.10.2019 15:30

Mathematics, 05.10.2019 15:30



Biology, 05.10.2019 15:30


Mathematics, 05.10.2019 15:30

Biology, 05.10.2019 15:30


Spanish, 05.10.2019 15:30



Mathematics, 05.10.2019 15:30



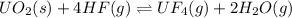
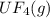 is added
is added

