
Chemistry, 27.03.2020 01:29 mcckenziee
When the oxide of generic metal M is heated at 25.0 ∘ C , a negligible amount of M is produced. MO 2 ( s ) − ⇀ ↽ − M ( s ) + O 2 ( g ) Δ G ∘ = 291.0 kJ mol When this reaction is coupled to the conversion of graphite to carbon dioxide, it becomes spontaneous. What is the chemical equation of this coupled process? Show that the reaction is in equilibrium. Include physical states and represent graphite as C ( s ) .

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 18:30
Asample of hydrated tin (ii) chloride (sncl2) has a mass of 4.90 g. when it is dehydrated, it has a mass of 4.10 g. which is the correct chemical formula for the hydrate? sncl2•2h2o sncl2•4h2o sncl2•6h2o
Answers: 2

Chemistry, 22.06.2019 21:30
If you burn 46.6 g of hydrogen and produce 416 g of water, how much oxygen reacted
Answers: 3

Chemistry, 22.06.2019 22:30
Which is a characteristic of the electron sea model for metallic bonding? molecular orbitals overlap to produce bands. electrons flow easily between metal nuclei. electrons are in fixed positions in the orbitals. atomic nuclei are arranged in an irregular pattern.
Answers: 3

Chemistry, 23.06.2019 00:30
How many moles of co2 are produced during the complete combustion of 3.6 moles of c2h6
Answers: 1
You know the right answer?
When the oxide of generic metal M is heated at 25.0 ∘ C , a negligible amount of M is produced. MO 2...
Questions

History, 11.09.2019 08:20

History, 11.09.2019 08:20


Mathematics, 11.09.2019 08:20

Biology, 11.09.2019 08:20


Medicine, 11.09.2019 08:20

History, 11.09.2019 08:20


English, 11.09.2019 08:20

History, 11.09.2019 08:20



Chemistry, 11.09.2019 09:10


French, 11.09.2019 09:10

Mathematics, 11.09.2019 09:10


Medicine, 11.09.2019 09:10

Mathematics, 11.09.2019 09:10

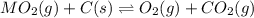
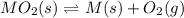 .....[1]
.....[1]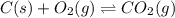 ..[2]
..[2]

