Chemistry, 26.03.2020 23:42 nadarius2017
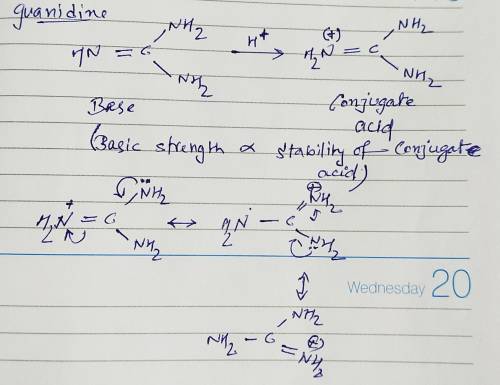
Guanidine is a stronger base than the typical amine. The increased basicity can be explained by drawing the resonance structures of the protonated guanidine. The protonated guanidine (A) has been drawn for you. Draw major resonance structures, one each in boxes B and C, and one minor resonance structure in Box D. Be sure to include the formal charge, lone pairs, and hydrogens on nitrogen for structures B, C, and D.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:00
If a plot weight (in g) vs. volume (in ml) for a metal gave the equation y= 13.41x and r^2=0.9981 what is the density of the metal?
Answers: 2

Chemistry, 22.06.2019 07:50
In which situation can a mixture always be called a solution
Answers: 3

Chemistry, 22.06.2019 11:50
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2

Chemistry, 23.06.2019 00:30
You are attempting to recrystallize a crude product mixture. you add the appropriate amount of hot solvent and are allowing the solution to slowly cool to room temperature. however, at room temperature no crystals have appeared, which of the following methods should be used to induce crystallization? choose all correct answers. a) place the flask in an ice bath. b) swirl the contents of the flask. c) add a small seed crystal of the desired product. d) scratch the inside of the glassware using a stir rod. it can be multiple choices
Answers: 3
You know the right answer?
Guanidine is a stronger base than the typical amine. The increased basicity can be explained by draw...
Questions



Spanish, 24.06.2019 11:00








Mathematics, 24.06.2019 11:00


Biology, 24.06.2019 11:00


Mathematics, 24.06.2019 11:00

Mathematics, 24.06.2019 11:00


Physics, 24.06.2019 11:00


History, 24.06.2019 11:00