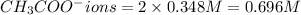In the laboratory you dissolve 14.8 g of chromium(II) acetate in a volumetric flask and add water to a total volume of 250 mL. What is the molarity of the solution? M. What is the concentration of the chromium(II) cation? M. What is the concentration of the acetate anion? M. Submit AnswerRetry Entire Group

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:30
Energy is released during which phase changes? check all that apply. boiling condensing depositing freezing melting subliming
Answers: 2

Chemistry, 22.06.2019 01:30
Aroller coaster car is traveling down a track at 22 m/s. the car has a mass of 2000 kg. what is the kinetic energy of the car? a) 22,000 j b) 968,000 j c) 484,000 j d) 44,000 j
Answers: 2


You know the right answer?
In the laboratory you dissolve 14.8 g of chromium(II) acetate in a volumetric flask and add water to...
Questions



English, 10.11.2020 18:50


World Languages, 10.11.2020 18:50



Spanish, 10.11.2020 18:50

Mathematics, 10.11.2020 18:50

Mathematics, 10.11.2020 18:50


Biology, 10.11.2020 18:50

SAT, 10.11.2020 18:50



Mathematics, 10.11.2020 18:50


Business, 10.11.2020 18:50

Medicine, 10.11.2020 18:50

Mathematics, 10.11.2020 18:50

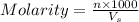
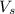 = volume of solution in ml = 150 ml
= volume of solution in ml = 150 ml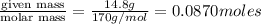
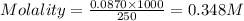
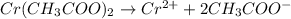
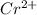 ions = 0.348 M
ions = 0.348 M