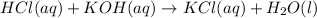During a laboratory activity, a student places 25.0 mL of HCl(aq) of unknown concentration into a flask. The student adds four drops of phenolphthalein to the solution in the flask. The solution is titrated with 0.150 M KOH(aq) until the solution appears faint pink. The volume of KOH(aq) added is 18.5 mL. Complete the equation for the neutralization reaction that occurs during the titration: KOH (aq) + HCl (aq) -->

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 11:00
An object becomes electrically charged when: electrons are created in it electrons from it are destroyed electrons are transferred to it protons from it are destroyed protons are created in it
Answers: 1

Chemistry, 22.06.2019 17:40
How much heat is added if 0.814g of water increase in temperature by 0.351 degree c?
Answers: 3

Chemistry, 22.06.2019 19:30
Describe the forces both attractive and repulsive that occur as two atoms move closer together.
Answers: 1
You know the right answer?
During a laboratory activity, a student places 25.0 mL of HCl(aq) of unknown concentration into a fl...
Questions

Mathematics, 21.02.2022 04:20



Social Studies, 21.02.2022 04:20


History, 21.02.2022 04:20



History, 21.02.2022 04:30

English, 21.02.2022 04:30



Mathematics, 21.02.2022 04:30

Mathematics, 21.02.2022 04:30


History, 21.02.2022 04:30

Computers and Technology, 21.02.2022 04:30



Mathematics, 21.02.2022 04:30