
Chemistry, 26.03.2020 20:59 makaylashrout77
Acetylene (C2H2) gas is often used in welding torches because of the very high heat produced when it reacts with oxygen (O2) gas, producing carbon dioxide gas and water vapor. Calculate the moles of acetylene needed to produce 0.090mol of carbon dioxide. Be sure your answer has a unit symbol, if necessary, and round it to 2 significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:30
Choose all the answers that apply. as ocean depth increases, temperature decreases temperature increases pressure increases pressure decreases salinity increases density increases
Answers: 2


Chemistry, 22.06.2019 12:30
If 22.5 liters of oxygen reacted with excess of hydrogen, how many liters of water vapor could be produced?
Answers: 3

Chemistry, 22.06.2019 21:00
Once similarity and one difference between a mixture of elements and a mixture of compounds
Answers: 3
You know the right answer?
Acetylene (C2H2) gas is often used in welding torches because of the very high heat produced when it...
Questions

World Languages, 11.10.2020 01:01



Mathematics, 11.10.2020 01:01





Mathematics, 11.10.2020 01:01

Mathematics, 11.10.2020 01:01



Social Studies, 11.10.2020 01:01


Biology, 11.10.2020 01:01


History, 11.10.2020 01:01

Mathematics, 11.10.2020 01:01


Mathematics, 11.10.2020 01:01

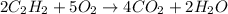
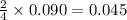 moles of acetylene
moles of acetylene

