
Chemistry, 26.03.2020 18:46 briannabrewer123
When a reaction has reached equilibrium... A. Q = K B. The rate of the forward reaction and the rate of the reverse reaction are equal. C. Both the forward and reverse reactions stop occurring. D. The concentration of products and the concentration of reactants are equal. E. The forward rate constant and the reverse rate constant are equal.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 00:40
During which time interval does the object travel approximately 10 meters
Answers: 3

Chemistry, 22.06.2019 01:40
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 23.06.2019 00:30
The molecular weight of carbon dioxide, co2, is 44.00 amu, and the molecular weight of nitrous dioxide, no2, is 46.01 amu, so no2 diffuses co2
Answers: 2
You know the right answer?
When a reaction has reached equilibrium... A. Q = K B. The rate of the forward reaction and the rate...
Questions

Mathematics, 16.07.2019 05:30

Mathematics, 16.07.2019 05:30


Mathematics, 16.07.2019 05:30



History, 16.07.2019 05:30


Business, 16.07.2019 05:30

Social Studies, 16.07.2019 05:30

Mathematics, 16.07.2019 05:30


Advanced Placement (AP), 16.07.2019 05:30


English, 16.07.2019 05:30



Mathematics, 16.07.2019 05:30

History, 16.07.2019 05:30

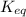 .
. cC
cC![K_{eq}=\frac{[C]^c}{[A]^a[B]^b}](/tpl/images/0565/3634/98de9.png)


