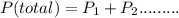Chemistry, 26.03.2020 16:46 manarsadi6
1. A sample of oxygen is collected over water at 22 ° C and 762 torr. What is the partial pressure of the dry oxygen? The vapor pressure of water at 22°C is 19.8 torr. A. 742 torr B. 782 torr C. 784 torr D. 750. Torr

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
How many moles are in 250 grams of tungsten (w)? * 4.4x10^23 moles 4.2x10^23 moles 0.7 moles 1.4 moles
Answers: 3

Chemistry, 22.06.2019 06:00
24. a sports ball is inflated to an internal pressure of 1.85 atm at room temperature (25 °c). if the ball is then played with outside where the temperature is 7.5 °c, what will be the new pressure of the ball? assume the ball does not change in volume nor does any air leak from the ball a) 0.555 atm b) 1.74 atm c) 1.85 atm d) 1.97 atm
Answers: 2

Chemistry, 22.06.2019 12:00
Explain what happens at the saturation point when adding salt to water at room temperature.
Answers: 1

Chemistry, 22.06.2019 23:30
If maltose undergoes hydrolysis what subunits does it results to?
Answers: 2
You know the right answer?
1. A sample of oxygen is collected over water at 22 ° C and 762 torr. What is the partial pressure o...
Questions



Mathematics, 30.06.2019 06:50

Spanish, 30.06.2019 06:50

Mathematics, 30.06.2019 06:50

Mathematics, 30.06.2019 06:50

English, 30.06.2019 06:50

English, 30.06.2019 06:50


Physics, 30.06.2019 06:50

English, 30.06.2019 06:50

Mathematics, 30.06.2019 06:50







Mathematics, 30.06.2019 06:50

English, 30.06.2019 07:00