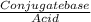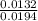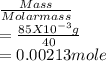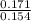Chemistry, 26.03.2020 05:19 kkjones1536
A 100.0-mL buffer solution is 0.175 M in HClO and 0.150 M in NaClO. Part A: What is the initial pH of this solution?Part B: What is the pH after addition of 150.0 mg of HBr?Part C: What is the pH after addition of 85.0 mg of NaOH?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 2

Chemistry, 22.06.2019 10:00
In a water molecule, hydrogen and oxygen are held together by a(an) bond. a) double covalent b) ionic c) nonpolar covalent d) hydrogen e) polar covalent
Answers: 1

Chemistry, 22.06.2019 11:00
When hydrochloric acid reacts with potassium hydroxide solution, the following reaction occurs. hcl (aq) + koh (aq) h2o (l) + kcl (aq) the reaction gives off heat energy, so it is an reaction.
Answers: 1

Chemistry, 22.06.2019 12:30
In france, grapes are 1.95 euros per kilogram. what is the cost of grapes, in dollars per pound, if the exchange rate is 1.14 dollars/euro? (2.6)
Answers: 3
You know the right answer?
A 100.0-mL buffer solution is 0.175 M in HClO and 0.150 M in NaClO. Part A: What is the initial pH o...
Questions


Mathematics, 14.07.2020 22:01



Mathematics, 14.07.2020 22:01


History, 14.07.2020 22:01



Mathematics, 14.07.2020 22:01

Mathematics, 14.07.2020 22:01



English, 14.07.2020 22:01



Mathematics, 14.07.2020 22:01


History, 14.07.2020 22:01

![\frac{[Conjugate base]}{[Acid]}](/tpl/images/0564/8323/225c1.png)
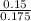
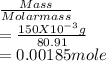
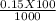 = 0.015
= 0.015 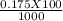 = 0.0175
= 0.0175