
Chemistry, 26.03.2020 04:28 jeffffffff
3.15 mol of an unknown solid is placed into enough water to make 150.0 mL of solution. The solution's temperature increases by 19.2°C. Calculate ∆H for the dissolution of the unknown solid. (The specific heat of the solution is 4.18 J/g・°C and the density of the solution is 1.20 g/mL).

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 15:10
Which statement describes the phase change that occurs when dry ice is placed in an open container at room temperature?
Answers: 1


Chemistry, 23.06.2019 00:00
How many peaks will be present in a mass spectrum for brcl?
Answers: 1

Chemistry, 23.06.2019 01:20
Use the de broglie's wave equation to find the wavelength of an electron moving at 4.5 × 106 m/s. show your work. note: h= plank's constant (6.62607 x 10-34 j s)
Answers: 1
You know the right answer?
3.15 mol of an unknown solid is placed into enough water to make 150.0 mL of solution. The solution'...
Questions

Social Studies, 16.07.2019 23:00

Mathematics, 16.07.2019 23:00



Biology, 16.07.2019 23:00

Mathematics, 16.07.2019 23:00



History, 16.07.2019 23:00




History, 16.07.2019 23:00

History, 16.07.2019 23:00

History, 16.07.2019 23:00




History, 16.07.2019 23:00

Mathematics, 16.07.2019 23:00

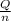 =
= 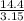 = 4.6 kj/mole
= 4.6 kj/mole

