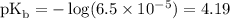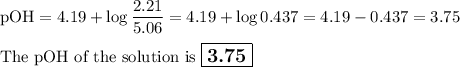Chemistry, 26.03.2020 03:19 danielle413
Calculate the pOH of a solution that results from mixing 46 mL of 0.11 M trimethylamine ((CH3)3N) with 17 mL of 0.13 M (CH3)3NHCl. The Kb value for (CH3)3N is 6.5 x 10-5.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:30
What are the major products produced in the combustion of c10h22 under the following conditions? write balanced chemical equations for each. a. an excess of oxygen b. a slightly limited oxygen supply c. a very limited supply of oxygen d. the compound is burned in air
Answers: 2

Chemistry, 22.06.2019 13:00
Jose and eric were given four samples in lab. the results of their analysis are shown in the table. based on the data they collected, which sample is most likely a metal?
Answers: 1

Chemistry, 22.06.2019 18:30
The number of moles of a given mass of a substance can be found without knowing its molecular formula or molar mass. true false
Answers: 1

You know the right answer?
Calculate the pOH of a solution that results from mixing 46 mL of 0.11 M trimethylamine ((CH3)3N) wi...
Questions



History, 14.11.2019 03:31


Mathematics, 14.11.2019 03:31





Social Studies, 14.11.2019 03:31

Business, 14.11.2019 03:31

Social Studies, 14.11.2019 03:31

English, 14.11.2019 03:31

Biology, 14.11.2019 03:31

English, 14.11.2019 03:31


Biology, 14.11.2019 03:31




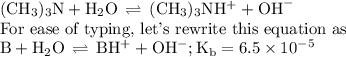
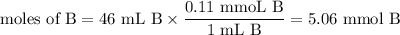
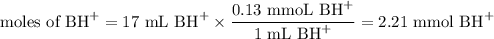
![\text{pOH} = \text{p}K_{\text{b}} + \log\dfrac{[\text{BH}^{+}]}{\text{[B]}}](/tpl/images/0564/6217/7ee6a.png)