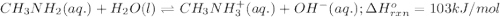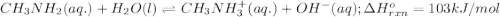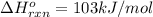Chemistry, 25.03.2020 22:37 kamjay2006
Use Le Châtelier's principle to predict how the equilibrium for the weak base methylamine responds to the indicated changes. CH 3 NH 2 ( aq ) + H 2 O ( l ) − ⇀ ↽ − CH 3 NH + 3 ( aq ) + OH − ( aq ) Δ H ∘ rxn = 103 kJ / mol

Answers: 2
Another question on Chemistry



Chemistry, 22.06.2019 13:00
Imagine that you push on a large rock. at what point does your effort change the rock’s mechanical energy?
Answers: 1

Chemistry, 22.06.2019 20:30
Water undergoes a large change in density at 0 ∘ c as it freezes to form ice. calculate the percent change in density that occurs when liquid water freezes to ice at 0 ∘ c given that
Answers: 2
You know the right answer?
Use Le Châtelier's principle to predict how the equilibrium for the weak base methylamine responds t...
Questions

History, 06.02.2022 07:20

Mathematics, 06.02.2022 07:20




History, 06.02.2022 07:20

Mathematics, 06.02.2022 07:20

Mathematics, 06.02.2022 07:20

Mathematics, 06.02.2022 07:20





Mathematics, 06.02.2022 07:20


Mathematics, 06.02.2022 07:20