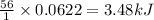Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:30
Aspirin has a density of 1.40 g/cm3 what is the volume in cubic centimeters of a tablet weighing 320 mg ?
Answers: 1

Chemistry, 22.06.2019 08:30
Which metal exist in liquid state and can be cut with knife ?
Answers: 2

Chemistry, 22.06.2019 22:10
Which aqueous solution of ki freezes at the lowest temperature? 1) 1 mol of ki in 500. g of water 2) 2 mol of ki in 500. g of water 3) 1 mol of ki in 1000. g of water 4) 2 mol of ki in 1000. g of water
Answers: 3

Chemistry, 22.06.2019 23:00
In the reaction h2co3 (aq) + 3nh3 (aq) = 2 nh4+ (aq) + co3 2-, how many electrons are transferred?
Answers: 3
You know the right answer?
The enthalpy of neutralization for the reaction of a strong acid with a strong base is -56 kJ/mol of...
Questions



Mathematics, 23.04.2021 04:30

Social Studies, 23.04.2021 04:30

Mathematics, 23.04.2021 04:30


Mathematics, 23.04.2021 04:30

Mathematics, 23.04.2021 04:30

History, 23.04.2021 04:30



Computers and Technology, 23.04.2021 04:30





Mathematics, 23.04.2021 04:30



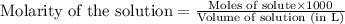 .....(1)
.....(1)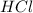 solution = 0.400 M
solution = 0.400 M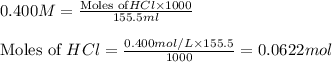
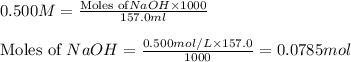

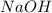
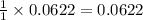 moles of
moles of