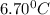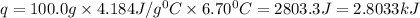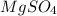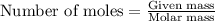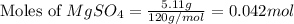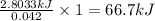Chemistry, 25.03.2020 05:26 makayla7635
5.11 g of MgSO₄ is placed into 100.0 mL of water. The water's temperature increases by 6.70°C. Calculate ∆H, in kJ/mol, for the dissolution of MgSO₄. (The specific heat of water is 4.18 J/g・°C and the density of the water is 1.00 g/mL). You can assume that the specific heat of the solution is the same as that of water.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:00
Ahydrogen atom has 1 electron. how many bonds can hydrogen form? a) 1 b) 2 c) 3 d) 4 e) 5
Answers: 3

Chemistry, 22.06.2019 16:30
4. a 20-kg child is tossed up into the air by her parent. the child is 2 meters off the ground traveling 5 m/s. circle one: ke / gpe / both show your work for finding the values of each type of energy the object has:
Answers: 1

Chemistry, 23.06.2019 03:00
A0.100-kilogram apple hangs in a tree 1.50 meter above the ground. ignore frictional effects, the total mechanical energy of the apples is
Answers: 1

Chemistry, 23.06.2019 05:00
Which of the following describes qualitative data? a) recording the temperature of a solid as it is warmed. b) noting the color of a solution as it is heated. c) measuring the volume of an object by water displacement. d) taking the mass of an object using a balance.
Answers: 2
You know the right answer?
5.11 g of MgSO₄ is placed into 100.0 mL of water. The water's temperature increases by 6.70°C. Calcu...
Questions








Engineering, 26.07.2019 20:10












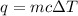
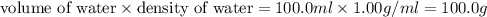
 = change in temperature =
= change in temperature =