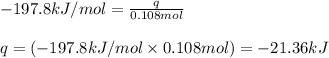Sulfur dioxide, SO 2 ( g ) , can react with oxygen to produce sulfur trioxide, SO 3 ( g ) , by the reaction 2 SO 2 ( g ) + O 2 ( g ) ⟶ 2 SO 3 ( g ) The standard enthalpies of formation for SO 2 ( g ) and SO 3 ( g ) are Δ H ∘ f [ SO 2 ( g ) ] = − 296.8 kJ / mol Δ Of [ SO3 ( g ) ] = − 395.7 kJ / mol Calculate the amount of energy in the form of heat that is produced when a volume of 2.67 L of SO 2 ( g ) is converted to 2.67 L of SO 3 ( g ) according to this process at a constant pressure and temperature of 1.00 bar and 25.0 °C . Assume ideal gas behavior.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:10
How many moles of gas are present if p=11 atm, v=12l, t=185k?
Answers: 1

Chemistry, 23.06.2019 03:30
How many grams of sodium chloride are in 250ml of a 2.5m naci solution
Answers: 1

Chemistry, 23.06.2019 05:00
Which of the following describes qualitative data? a) recording the temperature of a solid as it is warmed. b) noting the color of a solution as it is heated. c) measuring the volume of an object by water displacement. d) taking the mass of an object using a balance.
Answers: 2

You know the right answer?
Sulfur dioxide, SO 2 ( g ) , can react with oxygen to produce sulfur trioxide, SO 3 ( g ) , by the r...
Questions

Mathematics, 06.10.2019 23:30

Mathematics, 06.10.2019 23:30

Mathematics, 06.10.2019 23:30



Mathematics, 06.10.2019 23:30



Mathematics, 06.10.2019 23:30

History, 06.10.2019 23:30

Mathematics, 06.10.2019 23:30

Mathematics, 06.10.2019 23:30

History, 06.10.2019 23:30




Social Studies, 06.10.2019 23:30

Mathematics, 06.10.2019 23:30


Mathematics, 06.10.2019 23:30

![\Delta H^o_{rxn}=\sum [n\times \Delta H_f_{(product)}]-\sum [n\times \Delta H_f_{(reactant)}]](/tpl/images/0562/0323/e893d.png)
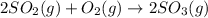
![\Delta H_{rxn}=[(2\times \Delta H_f_{(SO_3(g))})]-[(2\times \Delta H_f_{(SO_2(g))})+(1\times \Delta H_f_{(O_2(g))})]](/tpl/images/0562/0323/0e1fa.png)
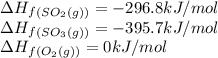
![\Delta H_{rxn}=[(2\times (-395.7))]-[(2\times (-296.8))+(1\times (0))]\\\\\Delta H_{rxn}=-197.8kJ/mol](/tpl/images/0562/0323/d0a68.png)
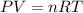
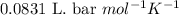
![25^oC=[25+273]K=298K](/tpl/images/0562/0323/df1f6.png)
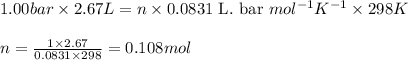
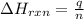
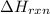 = enthalpy change of the reaction = -197.8 kJ/mol
= enthalpy change of the reaction = -197.8 kJ/mol