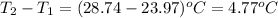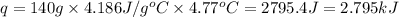Chemistry, 24.03.2020 22:28 2alshawe201
In a constant‑pressure calorimeter, 70.0 mL of 0.350 M Ba(OH)2 was added to 70.0 mL of 0.700 M HCl. The reaction caused the temperature of the solution to rise from 23.97 ∘C to 28.74 ∘C. If the solution has the same density and specific heat as water, what is heat absorbed by the solution? Assume that the total volume is the sum of the individual volumes. (And notice that the answer is in kJ).

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Asample of aluminum foil contains 8.60 × 1023 atoms. what is the mass of the foil?
Answers: 1

Chemistry, 22.06.2019 04:30
How much energy is made when a pice of wood burns. how do you know
Answers: 2

Chemistry, 22.06.2019 05:30
What happens to the atomic radius when an elctron is lost
Answers: 1

You know the right answer?
In a constant‑pressure calorimeter, 70.0 mL of 0.350 M Ba(OH)2 was added to 70.0 mL of 0.700 M HCl....
Questions

Chemistry, 22.03.2021 01:00



English, 22.03.2021 01:00





Computers and Technology, 22.03.2021 01:00

English, 22.03.2021 01:00


Mathematics, 22.03.2021 01:00

Geography, 22.03.2021 01:00




Mathematics, 22.03.2021 01:00

Chemistry, 22.03.2021 01:00

Biology, 22.03.2021 01:00

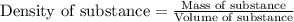
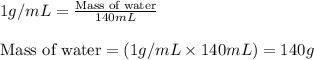
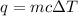
 = change in temperature =
= change in temperature =