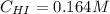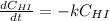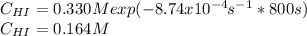Consider this reaction:
2HI(g) → H2(g)+ I2(g)
At a certain temperature it obeys this rate law.
Rate= 8.74 x 10^-4 s^1
Suppose a vessel contains HI at a concentration of 0.330M. Calculate the concentration of HI in the vessel 800 seconds later. You may assume no other reaction is important. Round your answer to significant digit

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 18:00
Chlorophyll a had the molecular formula c55h72mgn4o5 how many atoms are in this molecule
Answers: 2

Chemistry, 22.06.2019 20:30
A40 kilogram skier starts at the top of a 12 meter high slope. at the bottom, she is travelling 10 meters per second. how much energy does she lose to friction
Answers: 2

Chemistry, 23.06.2019 13:30
Why hydrochloric acid neutralized first when you titrate a mixture of hcl& ch3cooh against standard sodium hydroxide
Answers: 1

Chemistry, 23.06.2019 15:30
The gas in a sealed container has an absolute pressure of 9.25 atmospheres. if the air around the container is at standard pressure, what is the gauge pressure inside the container
Answers: 1
You know the right answer?
Consider this reaction:
2HI(g) → H2(g)+ I2(g)
At a certain temperature it obeys t...
2HI(g) → H2(g)+ I2(g)
At a certain temperature it obeys t...
Questions



Mathematics, 22.05.2020 02:57

Computers and Technology, 22.05.2020 02:57

Mathematics, 22.05.2020 02:57

Mathematics, 22.05.2020 02:57

Mathematics, 22.05.2020 02:57


Mathematics, 22.05.2020 02:57


Biology, 22.05.2020 02:57

History, 22.05.2020 02:57



English, 22.05.2020 02:57


Mathematics, 22.05.2020 02:57


Mathematics, 22.05.2020 02:57