
Chemistry, 24.03.2020 18:13 belladaviau1338
Iron(II) can be oxidized by an acidic K2Cr2O7 solution according to the net ionic equation: Cr2O72− + 6Fe2+ + 14H+ → 2Cr3+ + 6Fe3+ + 7H2O If it takes 35.5 mL of 0.0250 M K2Cr2O7 to titrate 25.0 mL of a solution containing Fe2+, what is the molar concentration of Fe2+ in the original solution?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 10:00
Nonpoint source pollution is difficult to control because it
Answers: 2

Chemistry, 22.06.2019 19:00
How many liters of ethylene glycol antifreeze (c2h6o2), with a density of 1.100 g/l, would you add to your car radiator containing 15.0 kg of water if you needed to protect your engine to - 21.5°c? for water, kf = 1.86°c m -1.
Answers: 1

Chemistry, 22.06.2019 21:30
What is the effect of returning nuclear reactor cooling water back into bodies of water?
Answers: 3

Chemistry, 23.06.2019 01:30
In which phase of mitosis do the spindle fibers pull the chromosomes apart to opposite sides of the cell ?
Answers: 1
You know the right answer?
Iron(II) can be oxidized by an acidic K2Cr2O7 solution according to the net ionic equation: Cr2O72−...
Questions



Health, 26.06.2019 03:00


Mathematics, 26.06.2019 03:00

History, 26.06.2019 03:00




Geography, 26.06.2019 03:00

History, 26.06.2019 03:00




Mathematics, 26.06.2019 03:00





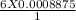 = 0.005325 mole of Fe²
= 0.005325 mole of Fe²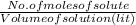
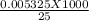 = 1.33 molar
= 1.33 molar


