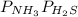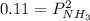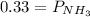Chemistry, 24.03.2020 16:55 ynclankaedon
Ammonium hydrogen sulfide NH4HS(s) decomposes on heating according to the reaction NH4HS(s) ↔ NH3(g) + H2S(g) At 25 °C the equilibrium cnstant of the reaction is Kp = 0.11. What is the total pressure at that temperature in a flask that was initially partially filled with a sizeable amount of NH4HS(s)?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:30
The big bang nucleosynthesis theory states that elements were produced in the first few minutes of the big bang while elements have their origins in the interiors of stars, forming much later in the history of the universe.
Answers: 1

Chemistry, 22.06.2019 10:00
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1

Chemistry, 22.06.2019 10:30
Great amounts of electromagnetic energy from our sun and other bodies in space travel through space. which is a logical conclusion about these electromagnetic waves? their energy must be very their frequency must be very low these waves can travel without a medium they only travel through a vacuum of space
Answers: 2

Chemistry, 22.06.2019 16:00
Sulfuric acid is a polyprotic acid. write balanced chemical equations for the sequence of reactions that sulfuric acid can undergo when it's dissolved in water.
Answers: 2
You know the right answer?
Ammonium hydrogen sulfide NH4HS(s) decomposes on heating according to the reaction NH4HS(s) ↔ NH3(g)...
Questions

Mathematics, 09.12.2021 02:10


Computers and Technology, 09.12.2021 02:10


Mathematics, 09.12.2021 02:10


Computers and Technology, 09.12.2021 02:10


Mathematics, 09.12.2021 02:10

English, 09.12.2021 02:10


Mathematics, 09.12.2021 02:10



Mathematics, 09.12.2021 02:10




Mathematics, 09.12.2021 02:10