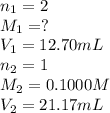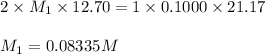Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Drag each label to the correct location on the chart. classify each reaction as endothermic or exothermic.
Answers: 1


Chemistry, 22.06.2019 21:50
Liquid from a brewery fermentation contains 10% ethanol and 90% water. part of the fermentation product (50,000 kg/h) is pumped to a distillation column on the factory site. under current operating conditions, a distillate of 45% ethanol and 55% water is produced from the top of the column at a rate of one-tenth that of the feed. what is the composition of the waste "bottoms" from the still?
Answers: 2

Chemistry, 23.06.2019 03:00
Abaker touches a pie right after taking it out of the oven. which statement best explains why the pie feels hot?
Answers: 1
You know the right answer?
A solution of malonic acid, H2C3H2O4 , was standardized by titration with 0.1000 M NaOH solution. If...
Questions

Social Studies, 01.10.2019 18:00


Health, 01.10.2019 18:00


Social Studies, 01.10.2019 18:00

Social Studies, 01.10.2019 18:00

History, 01.10.2019 18:00









Physics, 01.10.2019 18:00

Mathematics, 01.10.2019 18:00

Mathematics, 01.10.2019 18:00


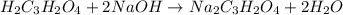
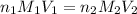
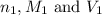 are the n-factor, molarity and volume of acid which is
are the n-factor, molarity and volume of acid which is 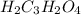
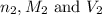 are the n-factor, molarity and volume of base which is NaOH.
are the n-factor, molarity and volume of base which is NaOH.