
A 18.08-g sample of the ionic compound , where is the anion of a weak acid, was dissolved in enough water to make 116.0 mL of solution and was then titrated with 0.140 M . After 500.0 mL was added, the pH was 4.63. The experimenter found that 1.00 L of 0.140 M was required to reach the stoichiometric point of the titration. a What is the molar mass of ? Molar mass = 129.14 g/mol b Calculate the pH of the solution at the stoichiometric point of the titration.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
What is the maximum amount of al2(so4)3 which could be formed from 15.84 g of al and 12.89 g of cuso4?
Answers: 2


Chemistry, 22.06.2019 05:50
Why doesn't heat added to water make the tempature rise above 100c
Answers: 2

Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1
You know the right answer?
A 18.08-g sample of the ionic compound , where is the anion of a weak acid, was dissolved in enough...
Questions

History, 02.06.2020 08:59

English, 02.06.2020 08:59

English, 02.06.2020 08:59

Business, 02.06.2020 08:59




Mathematics, 02.06.2020 08:59

Mathematics, 02.06.2020 08:59


Mathematics, 02.06.2020 08:59

Social Studies, 02.06.2020 08:59

Mathematics, 02.06.2020 08:59

Mathematics, 02.06.2020 08:59


Mathematics, 02.06.2020 08:59

Mathematics, 02.06.2020 08:59

English, 02.06.2020 08:59


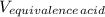 = 1.00 L
= 1.00 L
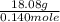
![10^{-4.63]](/tpl/images/0559/8018/9ece0.png)
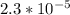
![[A^-]equ = \frac{0.140M*1.00L}{1.00L+0.116L}](/tpl/images/0559/8018/a9cf4.png)
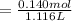
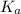 of HA =
of HA = 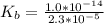
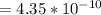
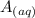 +
+ 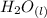
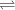
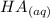 +
+ 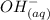
![K_b = \frac{[HA][OH^-]}{[A^-]}](/tpl/images/0559/8018/12da8.png)
![4.35*10^{-10} = \frac{[x][x]}{[0.1255-x]}](/tpl/images/0559/8018/ab7b7.png)
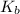 is very small, (o.1255 - x) = 0.1255
is very small, (o.1255 - x) = 0.1255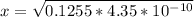
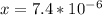
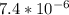 ]
]


