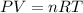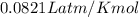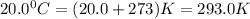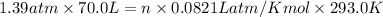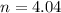Chemistry, 23.03.2020 20:39 negativechill
A 70.0-L steel tank at 20.0°C contains acetylene gas, C2H2, at a pressure of 1.39 atm. Assuming ideal behavior, how many grams of acetylene are in the tank? A 70.0-L steel tank at 20.0°C contains acetylene gas, C2H2, at a pressure of 1.39 atm. Assuming ideal behavior, how many grams of acetylene are in the tank? 4.04 g 105 g 12.9 g 1540 g

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 14:00
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3

Chemistry, 22.06.2019 18:30
Two people each hold the end of a rope and create waves by moving their arms up and down. this wave is best classified as a transverse wave because a) both the rope particles and the wave are moving in the same direction. b) the wave is moving up and down as the particles of the rope move horizontally. c) the wave is moving horizontally as the particles of the rope move up and down. eliminate d) the wave is moving in a parallel direction with the motion of the person's arms.
Answers: 3


Chemistry, 23.06.2019 06:30
Acompound has the molecular formula c3h8. which class of organic compounds does it belong to?
Answers: 2
You know the right answer?
A 70.0-L steel tank at 20.0°C contains acetylene gas, C2H2, at a pressure of 1.39 atm. Assuming idea...
Questions


Computers and Technology, 26.01.2021 20:10

History, 26.01.2021 20:10


Chemistry, 26.01.2021 20:10




English, 26.01.2021 20:10


Mathematics, 26.01.2021 20:10

Mathematics, 26.01.2021 20:10

Mathematics, 26.01.2021 20:10

Spanish, 26.01.2021 20:10

Social Studies, 26.01.2021 20:10

Physics, 26.01.2021 20:10

Mathematics, 26.01.2021 20:10


Mathematics, 26.01.2021 20:10

Chemistry, 26.01.2021 20:10