Chemistry, 23.03.2020 20:01 lpssprinklezlps
Suppose of iron(II) bromide is dissolved in of a aqueous solution of silver nitrate. Calculate the final molarity of bromide anion in the solution. You can assume the volume of the solution doesn't change when the iron(II) bromide is dissolved in it. Round your answer to significant digits.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:00
What forms when chemical reactions combine pollution with sunlight?
Answers: 1

Chemistry, 23.06.2019 06:20
Examine the false statement. compounds are the smallest unit of an element that occur most commonly in nature. select the rewording of the statement that is true. a: atoms are the smallest unit of an element that commonly occur in nature. b: molecules are the smallest unit of an element or compound that commonly occur in nature. c: molecules are the smallest unit of a compound that occur on the periodic table. d: compounds are the smallest unit of an element that occur on the periodic table
Answers: 1

Chemistry, 23.06.2019 10:30
Amethod of separation that employs a system with two phases of matter, a mobile phase and a stationary phase, is called
Answers: 2

Chemistry, 23.06.2019 13:30
Why hydrochloric acid neutralized first when you titrate a mixture of hcl& ch3cooh against standard sodium hydroxide
Answers: 1
You know the right answer?
Suppose of iron(II) bromide is dissolved in of a aqueous solution of silver nitrate. Calculate the f...
Questions


Arts, 10.10.2019 05:30




Computers and Technology, 10.10.2019 05:30



Biology, 10.10.2019 05:30



Business, 10.10.2019 05:30





Mathematics, 10.10.2019 05:30



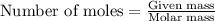
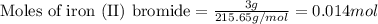
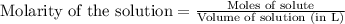 .....(1)
.....(1)
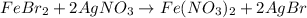
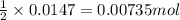 of iron (II) bromide
of iron (II) bromide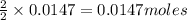 of silver bromide
of silver bromide