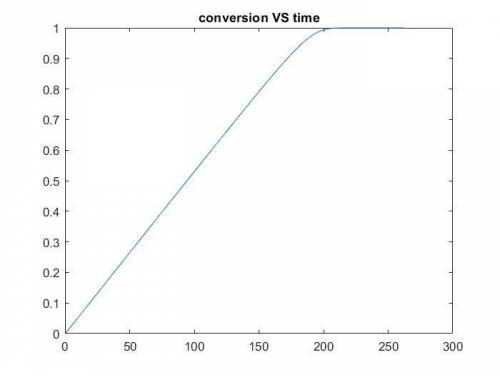
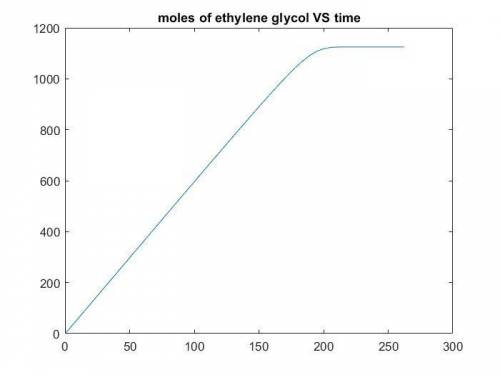
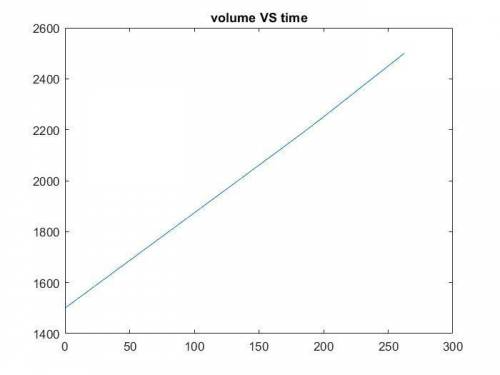
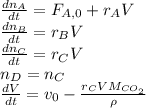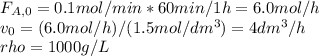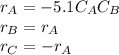The production of ethylene glycol from ethylene chlorohydrin and sodium bicarbonate ↑ is carried out in a semibatch reactor. A 1.5-molar solution of ethylene chlorohydrin is fed at a rate of 0.1 mole/minute to 1500 dm3 of a 0.75-molar solution of sodium bicarbonate. The reaction is elementary and carried out isothermally at 30°C where the specific reaction rate is 5.1 dm3/mol/h. Higher temperatures produce unwanted side reactions. The reactor can hold a maximum of 2500 dm3 of liquid. Assume constant density.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 22.06.2019 08:30
What method(s) do plants use to obtain nitrogen? select all that apply. absorb it from the atmosphere use bacteria to convert nitrogen to usable form obtain usable nitrogen compounds from the soil absorb nitrogen from water taken in at the roots
Answers: 3


Chemistry, 22.06.2019 19:00
Which statement best describes what happens when molecular compounds melt
Answers: 1
You know the right answer?
The production of ethylene glycol from ethylene chlorohydrin and sodium bicarbonate ↑ is carried out...
Questions

Mathematics, 17.03.2021 23:40

Mathematics, 17.03.2021 23:40



Mathematics, 17.03.2021 23:40

History, 17.03.2021 23:40

Mathematics, 17.03.2021 23:40



Mathematics, 17.03.2021 23:40

Mathematics, 17.03.2021 23:40

Mathematics, 17.03.2021 23:40

Mathematics, 17.03.2021 23:40


Mathematics, 17.03.2021 23:40


Mathematics, 17.03.2021 23:40


Mathematics, 17.03.2021 23:40

Mathematics, 17.03.2021 23:40