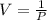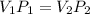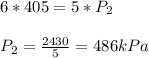Chemistry, 22.03.2020 22:59 LarryJoeseph
3. At a pressure of 405 kPa, the volume of a gas is 6.00 cm
temperature remains constant, at what pressure will the
a gas is 6.00 cm. Assuming the
at what pressure will the new volume be 4.00
cm? Written answer

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 06:30
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1

Chemistry, 22.06.2019 18:40
What is the binding energy of a nucleus that has a mass defect of 5.81*10-^29 kg a 5.23*10-^12 j b 3.15* 10^12 j c 1.57*10-3 j d 9.44*10^20 j
Answers: 1

You know the right answer?
3. At a pressure of 405 kPa, the volume of a gas is 6.00 cm
temperature remains constant, at w...
temperature remains constant, at w...
Questions

Mathematics, 19.10.2019 15:30



History, 19.10.2019 15:30

History, 19.10.2019 15:30


Mathematics, 19.10.2019 15:30

History, 19.10.2019 15:30

Mathematics, 19.10.2019 15:30


History, 19.10.2019 15:30

Geography, 19.10.2019 15:30


World Languages, 19.10.2019 15:30

Biology, 19.10.2019 15:30

Arts, 19.10.2019 15:30

Spanish, 19.10.2019 15:30

Health, 19.10.2019 15:30

Social Studies, 19.10.2019 15:30